WHO strongly advises against convalescent plasma for treating Covid patients
[ad_1]
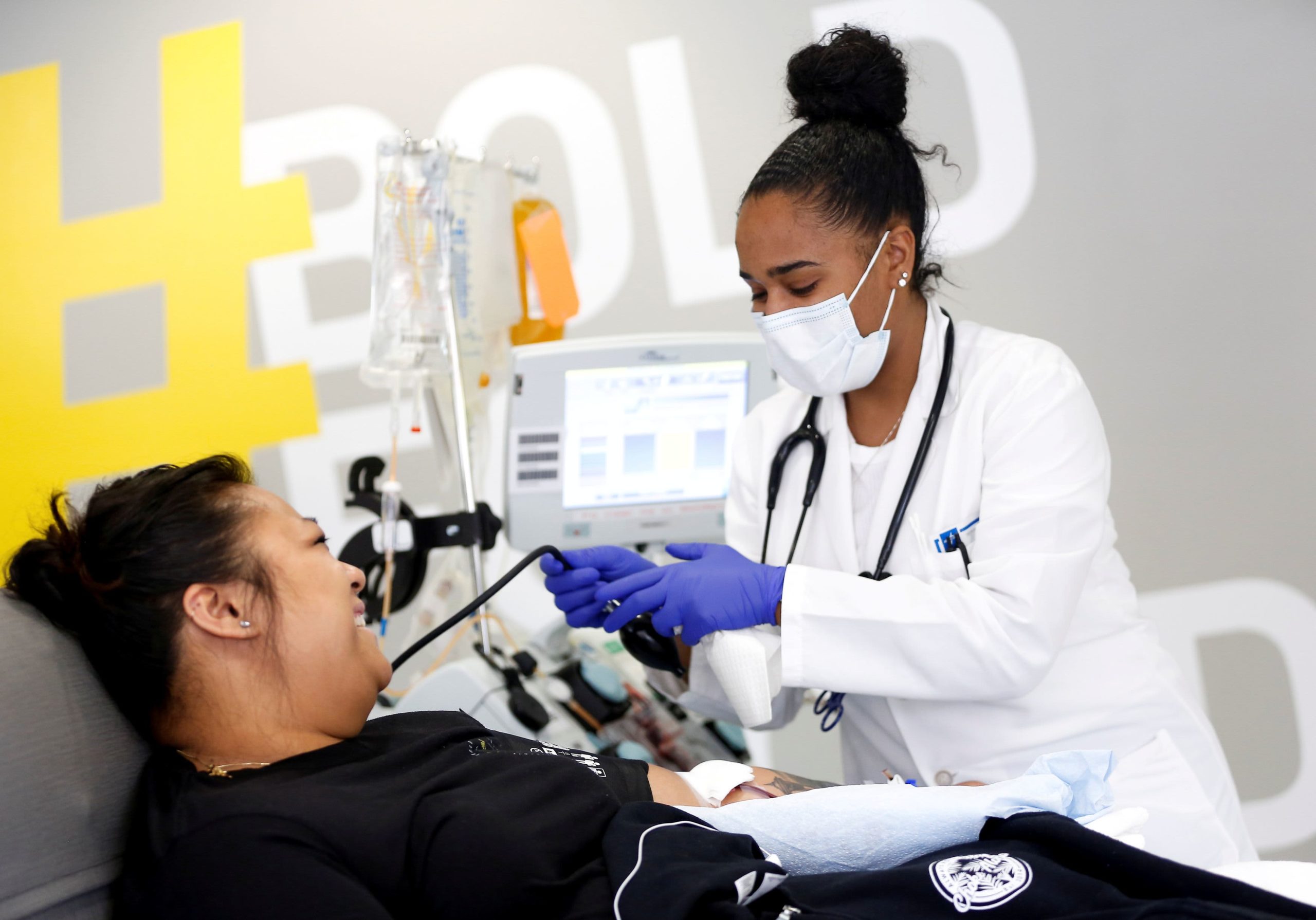
Kathryn Severson is a specialist in blood collection and holds a bag of convalescent Plasma from a patient who was treated for coronavirus at Central Seattle Donor Center of Bloodworks Northwest. This happened during the COVID-19 global epidemic, which occurred in Seattle on September 2, 2020.
Reuters| Reuters
Monday’s strong recommendation by the World Health Organization against convalescent plasma treatment for Covid-19 patients was based on research showing no improvements in those who have received it.
Convalescent plasma therapy is a form of plasma donation that involves blood from someone who has been cured. The plasma can then be transferred to a patient fighting the virus in the hopes the donor’s antibody will help.
The WHO guideline-development group concluded that there was not a clear benefit to critical outcomes like mortality or mechanical ventilation in patients with severe, non-severe or critical illnesses. They also found significant administrative costs and resource requirements.
According to the group, the treatment faces many practical problems, including finding donors and transporting plasma.
Based on 16 studies that involved more than 16,000 patients suffering from non-severe, severe or critical Covid infections, the recommendation has been made. Researchers should conduct randomized controlled trials to test the effectiveness of this treatment. This new recommendation was published in The British Medical Journal.
February 2012, U.S. Food and Drug Administration scaled back its convalescent plasma emergency use authorizationTo cover hospitalized patients at the beginning of disease progression, and for those with immune system disorders that prevent them from producing strong antibodies.
In its February emergency authorization, the FDA stated that “Plasma containing low levels of antibody has not been proven to be useful in COVID-19.”
When there was no alternative treatment, the original, more expansive authorization by the agency to all U.S. hospitalized patients was issued in August 2020. Alex Azar, Health and Human Services Secretary during the Trump Administration had described the “milestone achievement”, of the Convalescent Plasma being used at the time to counter Covid.
Two Covid treatment have been approved by the FDA since that time: GileadOctober 2020:’s Antiviral Drug Remdesivir RegeneronThe following month, he will be able to make his antibody cocktail. PfizerAbout a year ago, the two-dose vaccine for HIV was approved.
The National Institutes of Health in AugustConvalescent plasma was also found to not be beneficial in a study of 500 Covid adults at the University of Pittsburgh. This was also confirmed by the NIH. The trial was stopped in February due to its lack of effectiveness, the NIH said.
The New England Journal of Medicine.n a study published last monthThe convalescent plasma was not effective in preventing disease progression for high-risk patients when given one week after the onset of symptoms. The study also found that convalescent plasma did not have any clinical benefits for patients admitted to hospital late in their illness.
The study did show that convalescent plasma could reduce the progression of disease in outpatients older than 72 hours after symptom onset.
Pfizer MerckAre now requesting an emergency use authorization for antiviral oral drugs to decrease the likelihood of Covid hospitalization.
[ad_2]